Exploring the Atom: Printable Bohr Model Diagrams for Easy Learning
What is the Bohr Model?
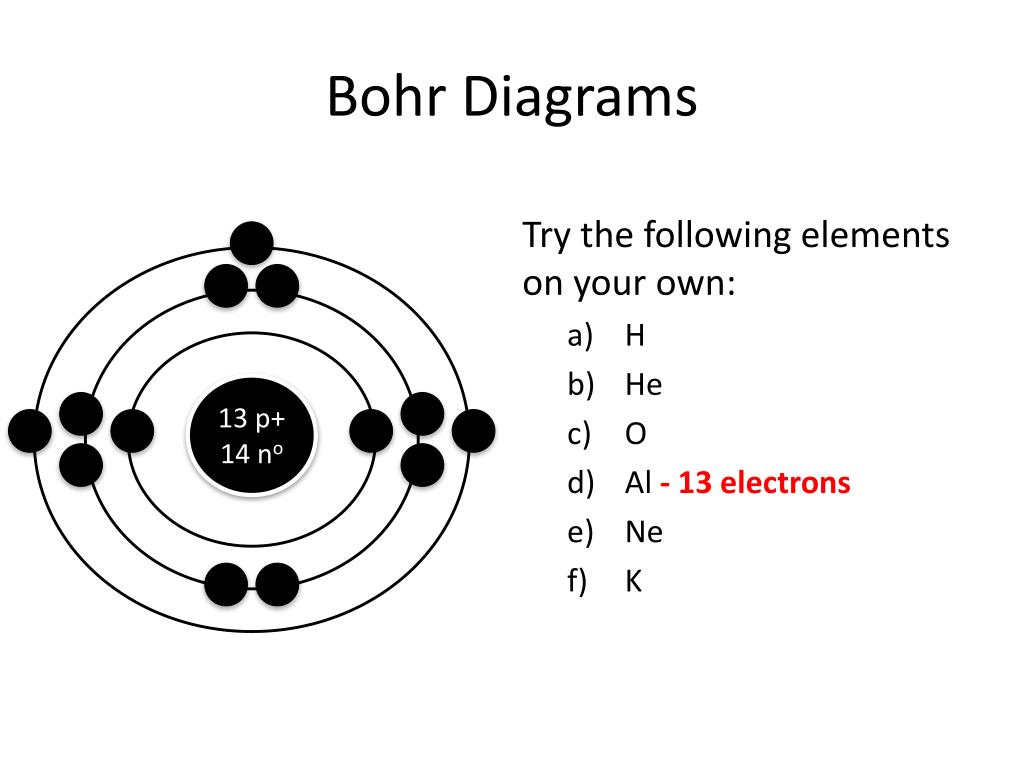
The Bohr model is a fundamental concept in chemistry and physics that helps us understand the structure of atoms. Developed by Niels Bohr, this model describes the atom as a small, positively charged nucleus surrounded by electrons in energy levels or shells. The Bohr model is a simplified representation of the atom, but it provides a clear and concise way to visualize the relationships between the nucleus and electrons.
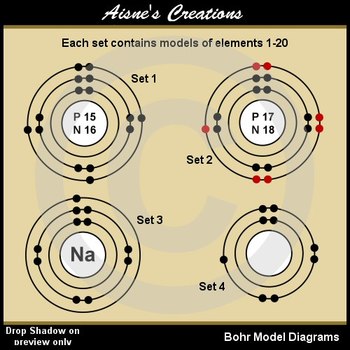
To better understand the Bohr model, it's essential to have a visual representation of the atom's structure. This is where printable Bohr model diagrams come in handy. These diagrams can be downloaded and printed, allowing students and educators to study and teach the Bohr model in a more engaging and interactive way. With printable diagrams, you can label and color-code the different parts of the atom, making it easier to identify and remember the various components.
Benefits of Printable Bohr Model Diagrams
What is the Bohr Model? The Bohr model is based on the idea that electrons occupy specific energy levels or shells around the nucleus. Each energy level can hold a certain number of electrons, and the electrons in each level have a specific amount of energy. The Bohr model also introduces the concept of electron spin and the idea that electrons can jump from one energy level to another by emitting or absorbing energy.
Benefits of Printable Bohr Model Diagrams Printable Bohr model diagrams offer several benefits for students and educators. They provide a hands-on way to learn about the atom's structure, allowing individuals to visualize and interact with the different components. Additionally, printable diagrams can be customized to fit specific learning needs, making them an excellent resource for teachers and students alike. By using printable Bohr model diagrams, you can gain a deeper understanding of the atom and its structure, making it easier to learn and appreciate the fundamental principles of chemistry and physics.